Hayley Abbiss1,2,3, John Moncur1, Scott J. Campbell1, Robert D. Trengove2,3
1SpectralWorks Ltd, United Kingdom; 2Separation Science and Metabolomics Laboratory, Murdoch University, WA, Australia; 3Metabolomics Australia, Western Australian Node, WA, Australia
First Published: ASMS 2017
Introduction
The introduction of the new EU Tobacco Product Directive will ensure that tobacco related products such as electronic cigarette liquids (e-liquids) will be tested to ensure compliance with the directive. Products may not contain more than 20 mg/mL of nicotine and no colouring or flavouring additives may be used. Additionally, vitamins or additives with a perceived health benefit have been prohibited and stimulants such as caffeine and taurine may not be added. Here we report a GC-MS method to profile e-liquids and describe how the software package AnalyzerPro® was used to perform quantitation and automatically create a library to search e-liquid products against to test for compliance with the directive.
Methods
Dilutions of e-liquid and a nicotine standard were prepared in water and isooctane. An aliquot of the water dilutions was dried and TMS-derivatised. Dilutions prepared in isooctane were transferred straight to vials for analysis. Samples were injected in split and splitless modes onto an Agilent 7200 series GCQTOFMS equipped with an Agilent 7693 auto injector and Agilent Factor Four VF-5ms capillary column. One microliter of sample was injected into the GC inlet, and analytes separated using a 24 minute temperature gradient, using helium as the carrier gas. Ionisation was by electron ionisation using a 70 eV electron beam and the mass analyser operated over the range m/z 45-600, acquiring at 10 Hz.
Nicotine standards were prepared in the concentration range 2.5-25 mg/mL. Standards and samples were diluted by a factor of 100 in isooctane and subsequently injected in split (50:1) mode.
Results
Data analysis revealed that a 1% solution of both water and isooctane dilutions of e-liquids injected with a split ratio of 50:1 was ideal to characterise the liquid profiles. Some saturation of the MS detector was observed. Higher sample concentrations and a split ratio of 20:1 introduced too much sample and resulted in significant chromatographic and MS overloading. This compromised data processing.
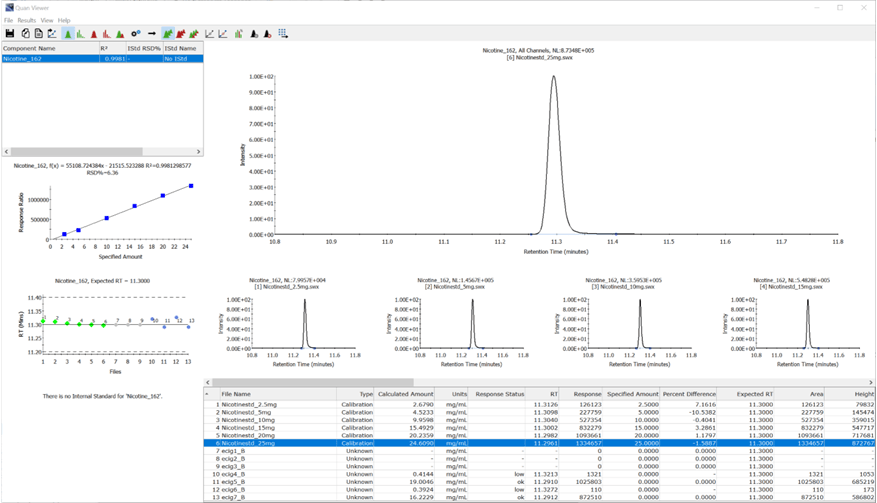
Figure 1. Quantitation results
A sample of vanilla flavoured e-liquid diluted with water and derivatised contained approximately 100 components including glycerol, cyclohexaneacetic acid and sugars such as tagatose, sorbose, mannose, galactose (best NIST matches) and sucrose. Two vanilla flavouring compounds were detected and vanilla flavouring compounds were also detected in the isooctane dilutions. The main component of the isooctane dilution was nicotine, its identity confirmed with an authentic standard and with a confident match to the NIST mass spectral database entry for nicotine.
There were fewer compounds observed in the water dilutions of a menthol e-liquid (approximately 70). Many of the compounds were artefacts of derivatisation or could not be identified. Glycerol, cyclohexaneacetic acid and threonic acid were identified. Menthol flavouring compounds and nicotine were detected in the isooctane dilutions of the liquid.
The chromatographic profile (Spectragram), for each of the e-liquids was created from the deconvolved components and automatically added to build a library of Spectragrams.
A proprietary matching algorithm based on the retention time, intensity and spectral quality of the components was used to compare each Spectragram against the others in the library.
Quantitation
Nicotine levels in each of the e-liquids were quantified against authentic standards. Each sample was labelled as containing 0 mg/mL nicotine. Figure 1 shows the quantitation report from AnalyzerPro and table 1 shows the results of the 7 samples tested, 4 had measurable levels of nicotine, 2 of which had levels approaching the upper limit of the EU Tobacco Product Directive.
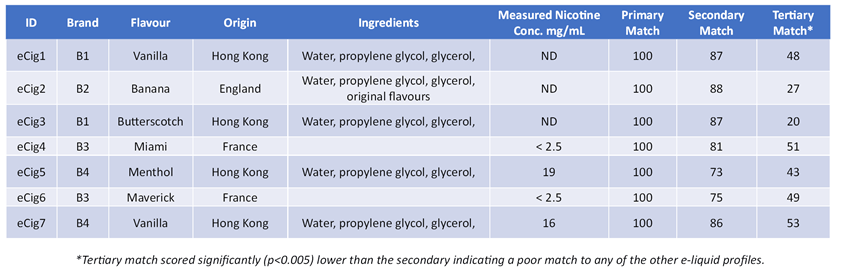
Table 1. Label information with measured nicotine (m/z 162) concentration and Spectragram matching results.
Qualitative Analysis
Spectragrams were created for each of the samples run in duplicate. The objective was to establish a proof of concept that these Spectragrams could be used to differentiate between the e-liquids on test. The matching algorithm used accounted for similarities and differences in both the chromatogram profile as well as the spectral quality of each of the components. A Spectragram comparison between replicate injections of the same e-liquid is shown in figure 2, showing a match confidence of 87%. The next best match is shown in figure 3, where the match value to a different e-liquid is 20%.
The match values for each of the e-liquid samples and their subsequent nearest match of the other test samples is shown in table 1. This shows significant differentiation between the e-liquid profiles.
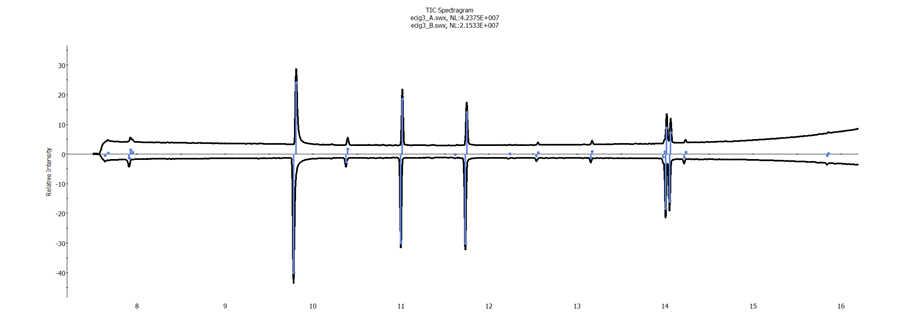
Figure 2. Spectragram of ecig3_A and ecig3_B, 87% match confidence.
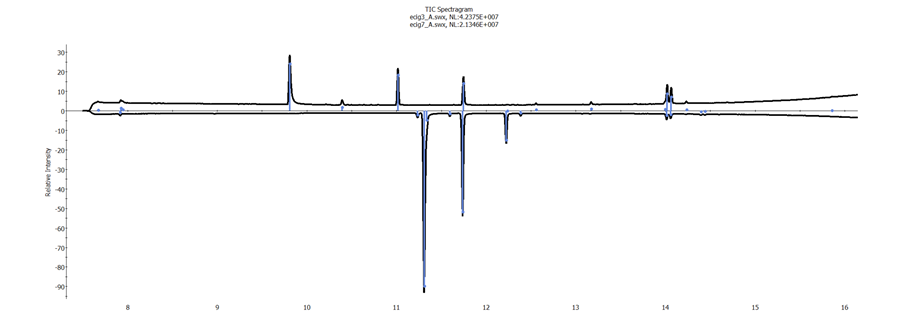
Figure 3. Spectragram of ecig3_A and ecig7_A, 20% match confidence.
Conclusions
Although the e-liquids used in this study were not sold as tobacco products, significant levels of nicotine were found in 4 of the 7 samples. The current EU Tobacco Product Directive does not apply to liquids that do not contain nicotine. This shows that regulation and control of e-liquid material is required for non-tobacco related products used in ‘Vaping’ with the health implications of exposure to known carcinogens. The law differs from country to country. In Western Australia, e-liquids containing nicotine are banned but there appears to be no formal process for checking liquids which are sold as not containing nicotine.
The comparisons using the Spectragram profile libraries of the samples showed highly significant differentiation between the test samples. In this proof of concept, the use of Spectragram profile analysis of e-liquid samples can differentiate samples from different sources.
The use of Spectragram profile matching worked well on a small number of samples and replicates. Further work is indicated to extend the Spectragram library database.
Acknowledgements
Metabolomics Australia is funded by the National Collaborative Research Infrastructure Strategy (NCRIS)