A metabolomics study of Plasmodium falciparum infection of red blood cells in the absence and presence of antimalarials
Nigel Deighton1, Joel Shuman1, Jianghong Qian1, Vladimir Shulaev1, Dominique Rasoloson2, Tosin Gisanrin2, Lirong Shi2, David Sullivan2 and Scott J Campbell3
1Virginia Bioinformatics Institute, Virginia Polytechnic Institute and State University, 1880 Pratt Drive, Blacksburg VA-24061-2 ; 2John Hopkins Bloomberg School of Public Health, John Hopkins University, Baltimore, MD-21205; 3SpectralWorks Ltd, The Heath Business & Technical Park, Runcorn, UK
First Published: ASMS 2004
Introduction
Malaria remains a scourge of the developing world, killing over a million people each year and infecting around 500 million [1]. Most victims live in sub-Saharan Africa, but the disease also afflicts Southeast Asia, South America and the Indian subcontinent. The situation has worsened over recent years as resistance has developed against chloroquine and sulphadoxine-pyrimethamine, the two drugs most commonly used to treat malaria [2]. The parasites are small ‘protozoan’ cells (the most prevalent species infecting humans are Plasmodium falciparum and P. vivax), which enter their human host through a mosquito bite. The emergence of drug resistance in the parasite necessitates the development of new antimalarial drugs, however their development is hindered by a lack of knowledge of the modes of action of existing compounds. For example, one mechanism believed to account for the toxicity of many of these drugs to the parasite is their ability to prevent hemozoin formation from the heme released during parasite degradation of hemoglobin. This mechanism has been questioned as it has been found that whilst chloroquine binds heme and prevents hemozoin formation, another potent drug, artemisinin also binds heme, but does not prevent hemozoin formation [3].
We are investigating the ability of a metabolomics approach to elucidate the mode of action of a number of known and potential antimalarial compounds of diverse structure.
Materials and Methods
Identical aliquots of erythrocytes infected with P. falciparum parasites (trophozoite stage) were harvested after incubation with drugs for three hours along with controls without drug at time zero and three hours. Samples were extracted by a Covaris E-100 acoustic transducer in a 400µl aliquot of 75% methanol containing four internal standards (ribitol, reserpine, 4-fluoro-3-nitrobenzoic acid, 3-fluorophenylalanine). GC-MS analysis was of derivatized (methoximated and trimethylsilylated) material (as in [5]), LC-MS/MS and CE-MS were on non-derivatized material.
CE-ESI-MS analyses were performed using a Beckman Coulter P/ACE MDQ capillary electrophoresis system interfaced to a Thermo LCQ DECA XP+ mass spectrometer. Separations were performed on a home made SMILE(+) 3-layer cationic-coated capillary [4] (80cm x 75µm). Separation electrolyte was 50mM ammonium acetate, pH8.5; co-axial sheath flow was 5mM ammonium acetate in 50:50 methanol:water (8 ?l/min). Separation voltage, 20kV, negative ion ESI, full scan, range 70-600m/z.
LC-MS/MS analyses were performed on a Thermo TSQ Quantum, electrospray positive ion. Analytes were separated on a 150x2mm Synergi Polar RP column (Phenomenex) with a linear gradient (30 mins) from 0.2% HCOOH to 70% acetonitrile. A single MS scan (100-1000m/z) was followed by two data-dependent scan events on the first and second most abundant ions in the MS scan event. Q2 conditions: 25V collision energy, 0.8mTorr argon.
GC-MS analyses were performed on a Thermo Trace DSQ, by a modification of [5]. A CombiPal autosampler injected 1 µl derivatized analytes into a 230°C injector with helium as the carrier and split and column flow at 25 and 1 ml·min-1, respectively. Chromatography was performed on a 60mx0.25mmx0.2 µm DB-5MS column (Agilent J&W): initial oven at 70°C for 5 min, followed by a 5°C ramp to 310°C for 6 min, and a post-run 70°C for 5 min. Ionization at 70eV with source at 200°C and positive full scan detection from 50-600 m/z at 2 scans·sec-1.
A variety of peak detection algorithms and front-end data reduction / extraction techniques including AMDIS, Thermo Xcalibur and SpectralWorks AnalyzerPro were used.
Identification of analytes was through searching custom-made MS (GC-MS) and MS/MS (LC-MS & CE-MS) libraries.
Results & Discussion
Analysis of the Covaris-extracted samples yielded complex multi-component chromatograms with each of the 3 analytical methods utilized.
Accumulation of lactate in P.falciparum-infected erythrocytes, produced through the fermentative metabolism of glucose, was evident, this accumulation being lower in all of the drug treatments (Figure 1). Additional distinctions between the sample treatments were evident from PCA analysis (example given in Figure 2).

Figure 1. Lactate accumulation in infected and treated-infected erythrocytes. Key: 1=uninfected, 2=infected, 3=cumene hydroperoxide, 4=neocuproine, 5=chloroquine, 6=artemisinin, 7=desferroxamine & artemisinin, 8=desferroxamine.
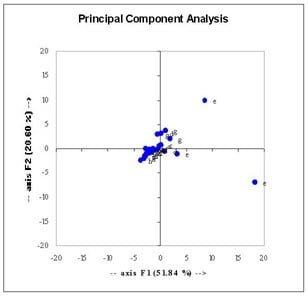
Figure 2. Principal component analysis of metabolite data. Key: a=uninfected, b=infected, c=artemisinin, d=chloroquine, e=dipicolinic acid, f=neocuproine, g=artemisinin & desferroxamine, h=cumene hydroperoxide.
Conclusions – Perspectives
We hereby report preliminary data of the metabolomics of Plasmodium falciparum-infected red blood cells through a concerted approach utilizing CE-MS, GC-MS and LC-MS techniques. Preliminary data obtained by this untargeted analytical approach highlights several distinguishing metabolite profiles from samples of uninfected, infected and infected (treated) erythrocytes.
Future method development is clearly necessary as it seems most unlikely that at present, we have achieved near complete coverage of the metabolome of the erythrocyte-parasite system. In addition, truly untargeted approaches, such as that described here, lead to two fundamental problems in metabolomics studies. Firstly, the prevalence of unidentified metabolites, and secondly, the higher than ideal limits of detection through the use of full-scan MS, where SRM/MRM would yield much greater sensitivity.
We are currently developing specific targeted analyses for the known metabolites of both human erythrocytes and Plasmodium falciparum.
Acknowledgement
This work was supported by the Virginia Bioinformatics Institute at Virginia Polytechnic Institute & State University and the John Hopkins Malaria Research Institute.
References
- Greenwood, B. & Mutabingwa, T. Nature 415, 670-672 (2002).
- Ridley, R. G. Nature 415, 686-693 (2002).
- Sullivan, D.J. et al., J.Biol.Chem. 273, 31103-31107 (1998).
- Latayama, H. et al. Anal.Chem. 70, 5272-5277 (1998).
- Fiehn, O. et al. Nat.Biotechnol. 18, 1157-1161 (2000).
The metabolism of P. falciparum
Carbohydrates
- Glucose uptake in an infected erythrocyte is 40-100 fold higher than the uninfected erythrocyte.
- Glucose is metabolized in a fermentation reaction yielding lactate.
- Some of the glycolytic intermediates may be shuttled into the pentose phosphate shunt to provide for nucleotide biosynthesis, and also provide NADPH.
Lipids
- The rapidly growing parasite requires large amounts of lipids for this increase in parasite surface area and volume of internal membranes.
- Historically, the parasite has been considered to be incapable of synthesizing fatty acids de novo and restricted to obtaining preformed fatty acids and lipids from the host.
- However, several enzymes associated with the type II fatty acid synthesis pathway have been identified in Plasmodium and appear to be located in the apicoplast.
Nucleotides and nucleic acids
- The malarial parasite obtains preformed purines by the salvage pathway and synthesizes pyrimidines de novo.
- The primary purine salvaged by the parasite is hypoxanthine which can be obtained from the host plasma A, G biosynthesis).
- The parasite cannot utilize preformed pyrimidines and must synthesize them from bicarbonate and glutamine.
Proteins
- Plasmodium hydrolyses hemoglobin, although most of the amino acids efflux the erythrocyte, only 16% of are incorporated into the parasite’s proteins.
- Plasmodium requires I, M, C, E, Q, P, Y (in vitro data). The parasite is also able to fix carbon dioxide and thereby synthesize A, D and E.
Hemoglobin breakdown
- Via plasmepsin produces large globin fragments (eg cleavage of F33-L34 bond of alpha-globin).
- Via falcipains and plasmepsin produces medium fragments of about 20 amino acid residues, then via falcilysin to produce small fragments of 6-8 residues.
- All of this occurs within the food vacuole, but amino acids are not produced here as the vacuole contains no exopeptidases. These are located within the parasite cytoplasm, suggesting efflux from vacuole to cytoplasm of small peptides.
